The 3DEP
3D Electrophysiology Platform
- Label-free electrophysiology
- Almost instant results
- Minimal preparation
- Easy operation
The 3DEP is the world’s first DEP cytometer, providing rapid, low-cost and label-free cell electrophysiology.
Insert cells into a disposable chip using a micropipette. Insert the chip into the reader. Close the door and hit the button. It’s that simple.
- The 3DEP analyses up to 20,000 cells simultaneously in 10 seconds.
- Low-cost disposable chips require less than 100uL and 100k cells.
- Measurements of membrane capacitance, surface conductance, and cytoplasm conductivity are instant. Resting membrane potential can be calculated from two measurements in different media.

The system is fast enough to allow real-time measurements of resting values – for example, for measuring ultradian rhythms. It is subtle enough to detect stem cell differentiation fate weeks before the same information can be detected by protein expression. It can detect what goes on inside a cell, for example during infection. It can detect early ionic events such as BK channel activity in processes such as apoptosis, much faster than by other methods.
The reason we say “electrophysiology is everywhere”, is that if you can measure electrophysiology this quickly and easily, you can use it as a marker for other things. The 3DEP can also discriminate between drug-resistant and drug-sensitive cells, or measure bacterial response to antibiotics and cancer cells to anti-cancer drugs – faster and more accurately than conventional methods such as MTT.
How does it work?
In order to analyse cells, the 3DEP uses a patented chip containing 20 “wells”, each carrying a number of electrodes down the inside wall. The electrodes in each well receive a specific frequency signal, allowing 20 frequencies (user-selectable, between 1kHz and 45MHz) to be analysed simultaneously. When the frequencies are applied to the wells, the cells inside the well move due to dielectrophoresis. If they experience “positive DEP” (that is, they are attracted to the electrodes) the cells move from the centre of the well towards the edge; if repelled from the electrodes they move towards the centre of the well. With a new cell type there is always scope for optimising the exposure time to produce the best fit (highest R) but we have found that in most cases, a 10 second exposure produces highly satisfactory results.
Diagnosis like you've never seen before.
Cells become diseased. Healthy cells become cancer cells. Blood cells become infected with malaria, macrophages with tuberculosis. Other diseases affect cell surface, others the membrane potential, others the morphology.
Electrophysiology changes faster than molecular biology. Cells beginning apoptosis change potassium levels almost instantly; caspases and other molecular markers follow hours behind. The 3DEP can determine IC50s ten times faster than MTT.
It’s not a surprise that cancer cells behave differently to normal, healthy cells at the same site. What is a surprise is that the differences include changes to the electrical signature of the cells.
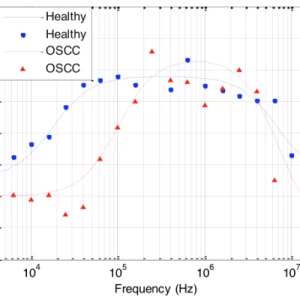
Oral cancer is in the top-ten most common cancers worldwide (in India it’s no.1), but survival rates haven’t improved in decades. Part of the reason is that cases aren’t identified early enough in primary care – but the time something definitely looks like cancer, it’s too late. On average, a visual inspection is about 70% accurate; what is needed is a quick, easy, low-cost and accurate test that doctors and dentists can use to identify those needing treatment at an earlier stage.

We used a 3DEP to tell the difference between brush samples taken from patients with oral cancer and healthy control subjects, using our MDV diagnostic marker. The 3DEP could identify cancer with 92% sensitivity, and identify healthy with 95% specificity (based on >100 participants).
Samples don’t need to be taken at point-of-care; they can be posted to a central lab for processing days after collection, keeping running costs down. This would work in the GP’s surgery or the dentist’s… even at home.

We have also used the same technique to detect bladder cancer, which is similar to oral cancer at a cellular level, but is the most expensive cancer to manage due to the need for lifelong, invasive monitoring for recurrence. When we applied our technique, we found very similar statistics. And there are other diseases that can be detected by this method – in fact, some that can only be detected by this method; we’re keeping some under wraps for now.

Your cells. Electrically.
Membrane Conductance
Membrane conductance describes the ability of a membrane to conduct charge, either through it or around it. It can be affected by the charges on the membrane surface, ion channel activity, and membrane morphology; the latter reason can be identified where there is a similar change in effective membrane capacitance, which is generally considered to be affected by membrane morphology almost exclusively. Multiplied by cell surface area, this gives the common whole-cell conductance parameter often used in cell electrophysiology.
Effective Membrane Capacitance
Effective membrane capacitance describes the ability of the membrane to store charge. Although in general the membrane composition changes little (and therefore the amount of charge a small patch of membrane can store remains approximately constant), the model assumes the surface is billiard-ball smooth and spherical. Deviation from this – such as when the cell surface is covered in blebs, microvilli or invaginations – means that there is actually more membrane than expected, meaning more charge is stored. This appears in the model as an elevated value of effective membrane capacitance per unit area of cell. Naturally, this makes the parameter a very good indicator of cell surface morphology; if a cell actually were perfectly spherical we would anticipate a capacitance per unit area of 8mFm-2, and a proportional increase indicates a similar proportional increase in area. Recent work has suggested that certain cell mechanisms altering the composition of the membrane (such as glycosylation) can have an effect without a morphology change, but the most common explanation is morphological. This has been a very effecting method of discriminating between cells – including observing changes in differentiating stem cells, or differences in cancerous vs. healthy cells. It is also useful for identifying morphology changes when interpreting changes in membrane conductance. As with conductance, multiplying by area gives the whole-cell capacitance commonly used in cell electrophysiology

Intracellular Conductivity
Intracellular conductivity is a measure of the ionic charge in the cytoplasm that can move freely and hence contribute to conduction. It is related to membrane potential; the cytoplasm conductivity is related to the sum of the positive and negative charges in the cell, whereas the membrane potential derives from the difference in numbers of positive and negative charge. As such it is a useful marker for change in ion concentrations, or ion channel activity. It is also a very useful marker for cell permeabilisation; when a cell membrane becomes permeable, the pores that appear allow connection between cell interior and exterior, causing a rapid drop in intracellular conductivity. It is also a useful marker for apoptosis, as one of the first events in the apoptotic cascade is ion efflux and water efflux, both of which can change the conductivity significantly (down in the former case, up in the latter). The actual change depends on the cell type and apoptosis-inducing agent, but the change is usually significant. If you divide 1 by the intracellular conductivity it gives the cytoplasm resistivity parameters commonly used in cell electrophysiology.
Multiple Populations
Sometimes a sample will contain a mixture of two or more cell types with radically different electrical characteristics. Where two or three such populations exist it is possible to model the two of them. For example, a common even in the study of apoptosis is the formation of apoptotic bodies, which typically appear as a second population with a radius of 0.5-1µm and a lower-than-normal intracellular conductivity. The ratio of the two populations is indicative of their relative concentration, although this is not a direct comparative measure (as the populations have different sizes and absorbances). We have successfully modelled up to three populations in the same sample, though this requires very high quality spectra, which can be obtained by averaging together many different spectra using the software provided. Where populations are very heterogeneous, such as in clinical samples taken from oral swabs, it is not possible to determine the properties of all of the populations; however, the 3DEP software can be used to discriminate between different samples on the basis of different dispersion characteristics. This technique has been used to classify clinical samples as cancerous or non-cancerous, where determination of the actual electrophysiological parameters is not required.
